Independent CRO · Berlin & Philadelphia · Est. 2006
Clinical trials where
imaging defines
the outcome.
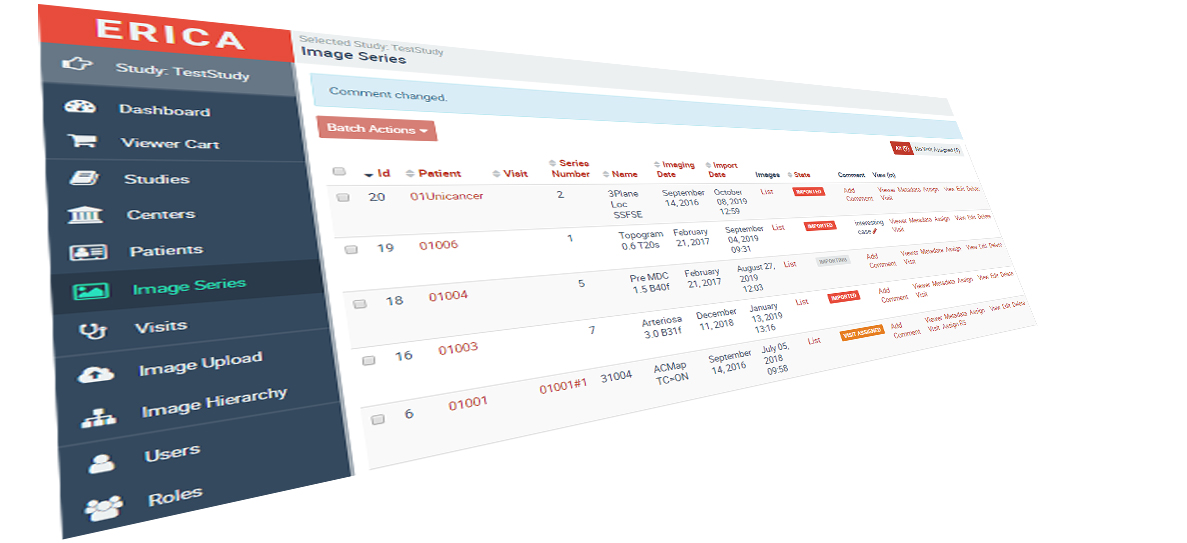
We specialise in studies where diagnostic imaging or radiopharmaceuticals define the outcome: contrast agent and diagnostic radiopharmaceutical development, RECIST response assessment in oncology, therapeutic radiopharmaceutical programmes, and medical device investigations. Imaging core lab, full CRO management, and consulting — across therapeutic areas and all development phases.